Recent Posts
THE STORY OF MY WILSON DISEASE: AGE 8 TO 53
By Barbara Noci I was born on the 6th of April 1972 in Empoli, a little city near Florence in [...]
Travis’s Story of Wilson Disease
Rare and Rural By Rhonda Rowland, WDA President We all love a small town. Images come to mind of holiday [...]
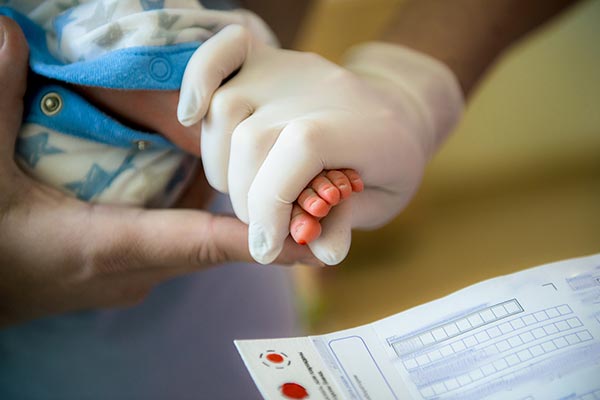
Newborn Screening for Wilson Disease
A 25-year dream coming close to reality By Alice Williams, WDA Communications Director Since the 1960s, the United States has [...]
Can Copper be Absorbed Through the Skin?
By Edward Tabor, MD, WDA Board Member The main source of copper for humans is food, and Wilson disease patients [...]
They Said “Yes” to Gene Therapy
By Rhonda Rowland, WDA PresidentWe continue the story of two trailblazers who are among the first Wilson disease (WD) patients [...]
A Makeover for an Old Drug
By Rhonda Rowland Wilson disease (WD) was always fatal until Dr. John Walshe, an English physician, discovered penicillamine in [...]
Registration and availability of Trientine hydrochloride – Australia
Important Announcement – Registration and availability of Trientine
dihydrochloride (AUST R 327984)
Waymade Australia Pty Ltd wishes to advise that Trientine Waymade (Trientine dihydrochloride) 250mg capsule bottle (x100) was registered with the Therapeutics Goods Administration on the 11th of January 2021.
The distribution of Trientine Waymade (Trientine dihydrochloride) 250mg capsule is managed by Clinect Pty Ltd.
Trientine Waymade is available for ordering by pharmacies in Australia, please find order and product enquiry information below;
For any enquiries contact Clinect:
Phone: 1800 899 005
Email: info@clinect.com.au
Stock is currently available to order via Symbion:
| Product Description | Item Code |
| TRIENTINE WAYMADE
(Trientine dihydrochloride) 250mg |
923753 |